Active Psoriatic Arthritis:
Active Psoriatic Arthritis:
Plaque Psoriasis:
Oral Ulcers Associated with Behçet’s Disease:






See before and after results by clicking here.
In a study of adults with mild to moderate plaque psoriasis,



See before and after results by clicking here.
In a study of adults with moderate to severe plaque psoriasis,
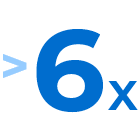
In a study of adults with moderate to severe plaque psoriasis and moderate to severe plaque psoriasis of the scalp,


More than half of people with psoriasis experience symptoms in their genital area at some point in their lives. This means itchy, cracked, dry skin in the places we feel it most: on the vulva, penis, scrotum, and the surrounding skin.
In a study of adults with moderate to severe plaque psoriasis and moderate to severe genital psoriasis,



See before and after results by clicking here.
In a study of adults with mild to moderate plaque psoriasis,




See before and after results by clicking here.
In a study of adults with moderate to severe plaque psoriasis,
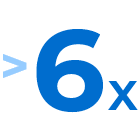
In a study of adults with moderate to severe plaque psoriasis and moderate to severe plaque psoriasis of the scalp,



More than half of people with psoriasis experience symptoms in their genital area at some point in their lives. This means itchy, cracked, dry skin in the places we feel it most: on the vulva, penis, scrotum, and the surrounding skin.
In a study of adults with moderate to severe plaque psoriasis and moderate to severe genital psoriasis,



more people saw reduced red/purple, flaky skin, so they could cover up less
In a study of mild to moderate plaque psoriasis, 22% of people taking Otezla achieved clearer skin after 4 months compared to 4% on placebo.

more people achieved clearer skin
In a study of moderate to severe plaque psoriasis, 33% of people taking Otezla achieved clearer skin after 4 months compared to 5% on placebo.

more people felt less itch to scratch
In a study of mild to moderate plaque psoriasis, 43% of people taking Otezla experienced less whole body itching after 4 months compared to 19% on placebo.

more people had less scalp psoriasis and left the hat at home
In a study of moderate to severe scalp psoriasis, 43% of people taking Otezla saw improvement after 4 months compared to 14% on placebo.



Did you know you may be eligible to pay as little as $0* out-of-pocket per month with the Otezla Co-Pay Program if you have commercial insurance? Your co-pay can be applied to deductible, co-insurance, and co-payment, and is available regardless of your income level.
First-time co-pay card user? Find out more.
No insurance? Learn more about accessing your Otezla prescription.
*For commercially insured patients only. Eligibility criteria and program maximums apply.
See AmgenSupportPlus.com/copay-terms for full Terms and Conditions.


Do not take Otezla 30 mg tablets or Otezla XR 75 mg extended-release tablets if you are allergic to apremilast or to any of the ingredients in Otezla/Otezla XR
Otezla/Otezla XR can cause serious side effects, including:
Some medicines may make Otezla/Otezla XR less effective and should not be taken with Otezla/Otezla XR. Tell your doctor about all the medicines you take, including prescription and non-prescription medicines.
The most common side effects include diarrhea, nausea, upper respiratory tract infection, tension headache, and headache. These are not all the possible side effects with Otezla/Otezla XR. Ask your doctor about other potential side effects. Tell your doctor about any side effect that bothers you or does not go away.
Tell your doctor if you are pregnant, planning to become pregnant or planning to breastfeed.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-332-1088.
Please click here for the Full Prescribing Information for Otezla/Otezla XR.
Prescription Otezla® /Otezla XRTM (apremilast) is used for the treatment of:
Active Psoriatic Arthritis:
Plaque Psoriasis:
Oral Ulcers Associated with Behçet’s Disease:
Do not take Otezla 30 mg tablets or Otezla XR 75 mg extended-release tablets if you are allergic to apremilast or to any of the ingredients in Otezla/Otezla XR.
Otezla/Otezla XR can cause serious side effects, including: